Answer: Patterning of the central nervous system is controlled by a set of inductive signals which control development of the neural tube through the regulation of transcription factors.
CNS patterning describes the process by which an ordered spatial arrangement is achieved in neural tissue. During embryonic development, three cell layers (referred to as germ layers) are formed in the embryo: the endoderm, mesoderm, and ectoderm. The endoderm gives rise to the lining of internal organs, while the mesoderm forms skeletal muscle, bone, and connective tissue. The ectoderm gives rise to hair, skin, and the entire nervous system. During the course of development, the ectoderm initially develops into a structure known as the neural plate, which eventually folds into the neural tube. This process by which the neural plate becomes the neural tube is known as neurulation and is depicted in the figure below.
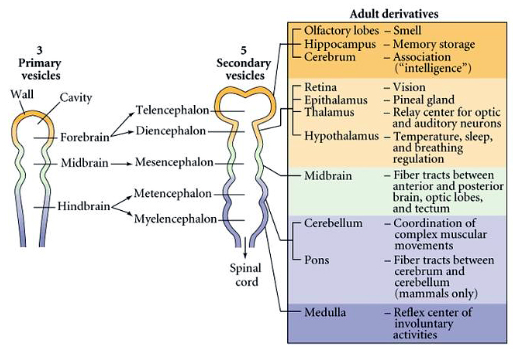
The neural tube then gives rise to three primary vesicles: the forebrain, midbrain, and hindbrain. The forebrain eventually differentiates into the telencephalon and diencephalon, while the hindbrain separates into the myelencephalon and metencephalon, as depicted in the figure below.
The question then arises, how does the neural tube give rise to its “adult derivatives”? The answer lies in the function of a number of inductive signals. A group of local signals determine fate of portion of the neural tube by regulating expression of transcription factors. This allows for the genetic control necessary for regionalization of both the dorsal-ventral and anterior-posterior axes of the nervous system.
The anterior-posterior and dorsal-ventral axes of the neural tube are patterned by different mechanisms. Signals from the endoderm and mesoderm contribute to the patterning of the neural plate along the anterior-posterior axis. This patterning can be attributed to specific signals, namely Wnt proteins, named after the Wingless and Int1 proteins found in Drosophila. At anterior levels, there is a decline of Wnt signaling, whereas posterior levels exhibit an increase in Wnt signaling, as shown in the figure below. Levels vary in between extremes, creating a gradient that allows for differentiation along the axis. The differentiation that occurs in response to this gradient is further refined by cell groups in the varying regions of the neural plate. Cells in the forebrain/midbrain regions express the transcription factor Otx2, while cells in the hindbrain region express transcription factor Gbx2, creating a point of transition between the midbrain and hindbrain, known as the isthmus. The isthmus functions as an organizer which expresses Engrailed transcription factors. A complex interaction between Wnt proteins, fibroblast growth factors, and Engrailed transcription factors stabilizes the midbrain-hindbrain boundary. While Wnt signals contribute to a gradient that gives rise to posterior specification, other factors such as fibroblast-growth-factors and retinoic acid also contribute to posteriorization. In the anterior portion of the neural tube, Wnt agonists such as cerberus dickkopf are secreted, contributing to anteriorization of the neural tube. Once this regionalization is achieved, signals in the anterior regions are secreted for further regionalization of the forebrain into the telencephalon and diencephalon.
The dorsal-ventral axis of the neural tube has inductive factors that contribute to patterning in a similar way to the rostrocaudal axis. These factors include sonic hedgehog (Shh) protein and bone morphogenic protein (BMP), which are secreted from secondary structures. The notochord establishes a secondary structure referred to as the floor plate, and the epidermis gives rise to one known as the roof plate. The secretion of Shh from the floor plate contributes to ventralization and opposes BMP signals from the roof plate; BMP contributes to dorsalization and opposes Shh signals from the floor plate. Both Shh and BMP are heavily concentrated at their corresponding secondary structures and decrease in concentration as they approach the medial portion of the tube. This gives rise to a concentration gradient where each point along the dorsal-ventral axis expresses different levels of Shh and BMP proteins. Any given horizontal section of the neural tube can be divided into domains of progenitor cells, each of which corresponds to a specific set of transcription factors (see figure below). The varying amounts of Shh and BMP signals at each location work to control the expression of these transcription factors in the neural tube. This mechanism of dorsoventral patterning is conserved along the extent of the neural tube.
Thus, the neural tube acquires its specification by utilizing inductive factors to control gene expression across the dorsal-ventral and anterior-posterior axes.